VISION & MISSION
Our ambitious goal is to develop a safe and efficient cell therapy based on genome-edited T cells with redirected specificity to sustainably combat IgA nephropathy (IgAN) – the most common glomerulonephritis and one of the most common causes of end-stage renal disease with unmet medical need.
Our specific cell therapy approach is also suitable for other diseases with selective B-cell pathogenesis, such as IgA myeloma and rheumatoid arthritis, but as a blueprint also for diseases of other Ig classes (e.g. IgG4).
Our novel concept offers a specific form of immunosuppression via Ig-(sub)class targeting redirected T cells in autoimmune diseases. Methodically, we benchmark three promising genome editing technologies, develop new standards for safety assessment and preclinical performance evaluation.
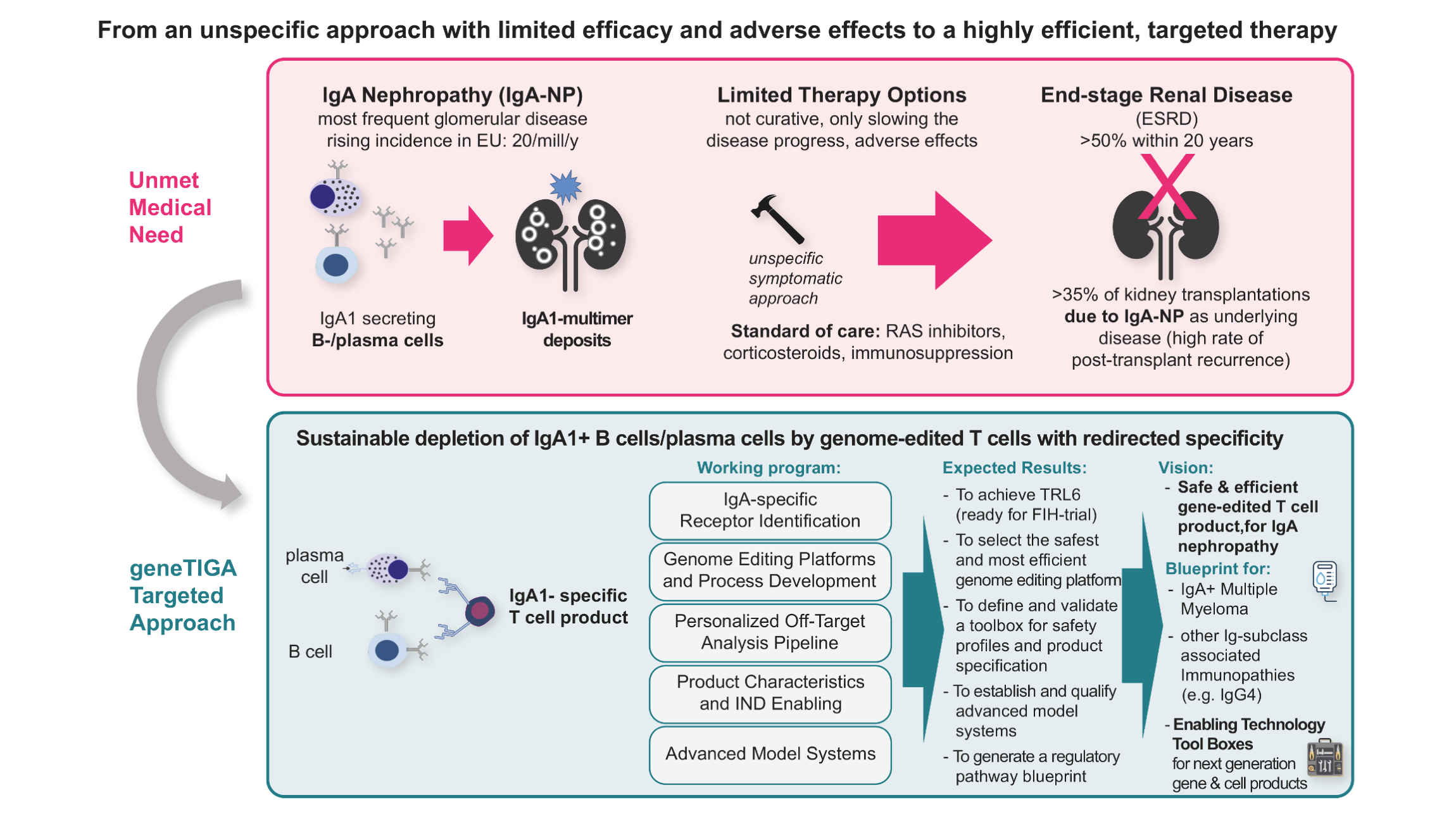
At the end we will have a lead candidate of a new “living drug” product envisioned as a one-time treatment for IgAN and other IgA-associated that will be ready to enter clinical FIH trials (entry into TRL6). In addition, geneTIGA delivers enabling technology toolboxes with exploitation options beyond of the core project. The aim is to reduce risks and accelerate the development of next-generation gene and cell products in general.
The project has thus, besides its scientific value, a high impact not only on the affected patients with IgAN and related immune diseases, but also for the European society by reducing the health economic burden caused by progressive chronic kidney disease, as well as by triggering innovation and business options in Europe´s biotech and pharma field.
